Sieweke Group
Our research is at the interface of immunology and stem cell research, with a main focus on macrophages. These cells play an important role in immunity and tissue regeneration. What happens inside and around the cell to differentiate from a hematopoietic stem cell into a macrophage, and what are the molecular and cellular mechanisms of self-renewal? We want to get to the bottom of these questions….
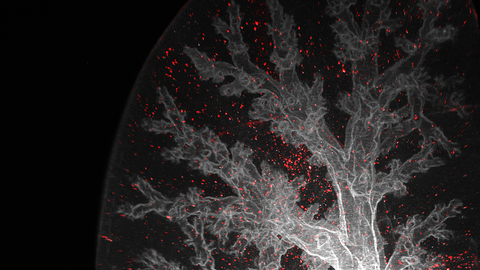
 © Sethuraman Subramanian
© Sethuraman Subramanian
Our research
Our recent findings lay the groundwork for new cellular therapy approaches in regenerative medicine.
One important line of our work has revealed mechanisms how hematopoietic stem cells can generate more myeloid cells, including macrophages, to protect transplant recipients from lethal infections.
Furthermore, we discovered that macrophages, mature cells of the immune system, could activate a network of self-renewal genes shared with embryonic stem cells that enables them to proliferate indefinitely. This makes it possible to amplify macrophages in culture as mature differentiated cells, without stem cell intermediates or tumorigenic transformation. Our results show that not only stem cells but also mature cells like macrophages can activate self-renewal mechanisms. This opens the door for new macrophage-based therapies.