Albert Group
The neocortex is considered to be the seat of higher cognitive functions in humans. During its evolution, most notably in humans, the neocortex has undergone considerable expansion, which is reflected by an increase in the number of neurons. Neocortical neurons are generated during development by neural stem and progenitor cells. Epigenetic mechanisms play a pivotal role in orchestrating the behavior of stem cells during development. We are interested in the mechanisms that regulate gene expression in neural stem cells, which have implications for our understanding of neocortex development and evolution, neural stem cell regulation and neurodevelopmental diseases.
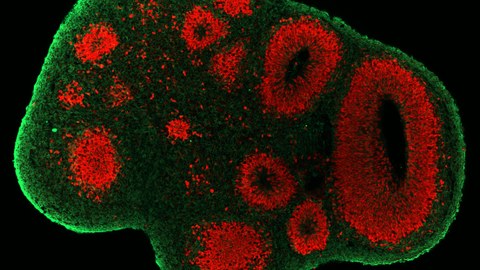
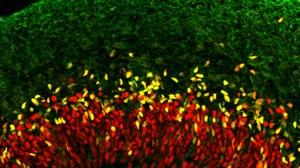
Figure: Neural progenitor cells of the developing neocortex. During neurogenesis, neural progenitor cells (SOX2/red; TBR2/yellow) give rise to neurons (TUJ1/green). Image by Theresa Schütze.
 © Theresa Schütze
© Theresa Schütze
Future projects and goals
The aims of our research are
- to elucidate the epigenetic regulation of human neocortex development
- to further develop models of neurodevelopmental disorders caused by mutations in epigenetic factors
- to understand how epigenetic mis-regulation contributes to neuropathologies and whether epigenetic changes and phenotypes can be reversed
- to identify and characterize gene regulatory regions that may have contributed to human brain evolution
Figure: Neural progenitor cells of the developing neocortex. During neurogenesis, neural progenitor cells (SOX2/red; TBR2/yellow) give rise to neurons (TUJ1/green). Image by Theresa Schütze.